
At a conference in honour of Dr Árpád Pusztai, Prof Vyvyan Howard exposed ongoing regulatory failure over GM crops and foods. Report: Claire Robinson
On 26-27 May, I attended a two-day conference in Edinburgh called The Integrity of Science, about the conflicts between corporate interests, public policy, and independent science, in honour of the late Dr Árpád Pusztai (1930–2021).
In the late 1990s Dr Pusztai carried out important experiments to test the safety of GMO potatoes and found that they harmed the health of rats. In 1998 he went public with his findings, saying on British TV that he would not eat GM potatoes and thought it was "very unfair to use our fellow citizens as guinea pigs" for GMO technology.
For his pains, he was fired from his institute and gagged from speaking about his research, which was later peer-reviewed and published in The Lancet. The potatoes were destroyed and the experiments never followed up. So scientists still do not know why the GM potatoes unexpectedly proved to be toxic. (The definitive account of the whole extraordinary drama of Pusztai's groundbreaking research and the attempts to destroy his reputation is described by the author and investigative journalist Andy Rowell in two carefully referenced chapters of his book, Don't Worry, It's Safe to Eat, republished with Andy Rowell's permission by GMWatch.)
This conference was a memorial and tribute to this great scientist and humanitarian, who stood up for scientific integrity against massive pressure from the scientific establishment and corporate interests. The presentations, by a stellar lineup of international speakers, looked at the state of scientific integrity in history and now. Shockingly, things have got no better since Dr Pusztai had his career destroyed, and in some ways they are worse.
 We will summarise some of the presentations at this important conference. We begin by covering the presentation by the toxico-pathologist Vyvyan Howard, Emeritus Professor at the University of Ulster.
We will summarise some of the presentations at this important conference. We begin by covering the presentation by the toxico-pathologist Vyvyan Howard, Emeritus Professor at the University of Ulster.
"Risk assessment and 'fact-free models'"
In his presentation, titled "Risk assessment and 'fact-free models'", Prof Howard explained what was wrong with the system of risk assessment. In particular, he exposed gaping holes in the risk assessment of GM crops and foods. He pointed out that he had originally written his presentation over 20 years ago and had planned to update it for the conference, but to his dismay he didn't have much to do, as the failures that he pointed out two decades ago still apply.
We should point out here that while we at GMWatch have long drawn attention to the shortcomings of GMO and pesticide risk assessment, we believe that it's far better to have even an inadequate risk assessment than none at all. Pretty much none at all is what the UK government plans for new GMOs that the developer claims are equivalent to conventionally bred organisms. And the risk assessment process – and the selection process for those doing the assessment – need to be reformed from the bottom up, rather than abandoned altogether for reasons of commercial expediency.
Prof Howard proposed ways this reform could be achieved. But under the current system as it is practised, as he showed, risk assessments cannot be assumed to prove the safety of any substance, GMOs included.
He began by emphasising the importance of the precautionary principle in GMO safety assessment, citing this definition: "When an activity raises threats of harm to human health or the environment, precautionary measures should be taken, even if some cause and effect relationships are not fully established scientifically."
Prof Howard said that risks from GM foods are potentially far greater than those from pharmaceutical drugs. That's because with a drug, unlike with a GMO food, the patient takes it by choice with their informed consent, in small amounts, usually for a limited period, and the effects are monitored.
In contrast, a GMO food could be consumed over a lifetime in relatively larger quantities. And if it's not labelled, shoppers have no choice over whether they expose themselves to it.
While GMO proponents are fond of claiming that Americans have been eating GMOs for decades with no ill effects, Prof Howard pointed out that no baseline data ("Where did we start from?") or exposure data ("Who has eaten what?") has been collected, so no one knows what effects there may have been. And if the harmful effect caused by a GM food were an increase in a common disease, it won't be noticed, as it will be buried in the general morass of disease. The only thing that could be deduced is that no one seems to have dropped dead from acute toxicity on eating a GM food.*
Problems with risk assessment
Prof Howard warned that risk assessment is "The main tool used to stop the implementation of the precautionary principle" and added that it is widely misunderstood, including by decision makers, as "proof" that technologies are safe.
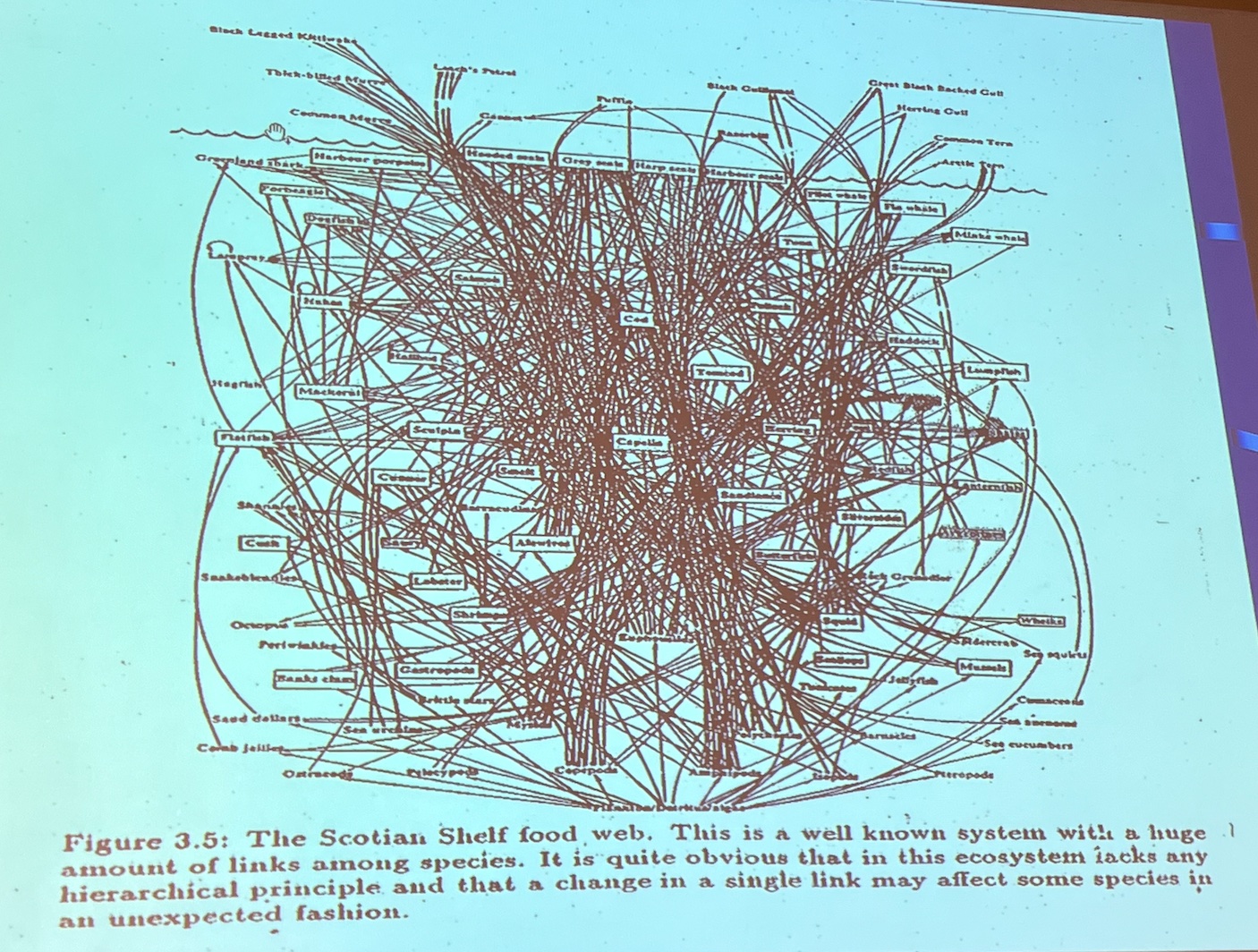
Despite years of experience and data gathering, unpredictable events still happen, showing that the hazard identification or hazard assessment steps of risk assessment simply aren't working. Given the immense complexity of biological systems (see slide below), that's not surprising. A former director of the US EPA warned, "We should remember that risk assessment can be likened to the captive spy: If you torture it long enough, it will tell you anything you want to know."
 An example of the failure of risk assessment is that all people were assumed to have had no appreciable prior exposure to dioxin and that therefore it was safe for them to receive a new pollution burden. Also, no measurements of actual body burdens of dioxin were presented in exposure assessments, despite the fact that taking such measurements would have been easy and it is already known that everyone on the planet has been exposed to dioxins.
An example of the failure of risk assessment is that all people were assumed to have had no appreciable prior exposure to dioxin and that therefore it was safe for them to receive a new pollution burden. Also, no measurements of actual body burdens of dioxin were presented in exposure assessments, despite the fact that taking such measurements would have been easy and it is already known that everyone on the planet has been exposed to dioxins.
With GMOs, there are several potential areas of risk, including genetic instability, horizontal gene transfer (e.g. antibiotic resistance genes – frequently used in genetic engineering – transferring to bacteria in the gut), and unintended effects such as toxicity and allergenicity. GMOs are assessed via a "test" of substantial equivalence, which only looks at a restricted number of substances in the GMO and can't effectively predict allergenicity or unpredicted toxicity.
Chardon LL
Prof Howard showed examples of risk assessment data provided to the regulator by companies to try to get their GMOs authorised. For each type of risk named, the developer had asserted that it was "low", "negligible", or "effectively zero" – without providing data to show that. In 2000 Aventis applied to the UK authorities to add its GM Chardon LL forage maize to the UK national seed list for commercial cultivation. It already had EU approval, which included the UK, but the crop needed to be added to the seed list before it could be sold to farmers.
The data that Aventis submitted to the authorities were completely inadequate, as Prof Howard showed. Though the GM maize was intended to feed cattle, only the purified PAT protein, which confers tolerance to the herbicide glufosinate ammonium, was isolated from canola – an irrelevant crop as the protein will express differently in maize. It was then fed to rats – an irrelevant species that cannot tell us if the GM maize was safe to feed to cattle, due to their different digestive systems. No feeding trial was done in cattle with the whole maize.
GMWatch adds that in other company tests, the GM maize was also fed to chickens. Concerns were raised about "suspicious" higher death rates among chickens that ate GM maize – twice as many chickens died, compared with those fed on non-GM maize. But despite Aventis's "fact-free" application, the UK government judged the GM maize to be safe.
Fortunately, in the face of heavyweight opposition, in 2004 Aventis (which had been bought out by Bayer) withdrew its application to commercialise the maize in the UK. But the withdrawal owed nothing to the regulatory risk assessment.
Royal Society mobilised against Dr Pusztai
 Prof Howard reminded the audience of the pressure on the scientific establishment to suppress inconvenient findings. In 1999 Richard Horton, editor of The Lancet, told the Guardian that he had been pressured from "all quarters", notably from the UK Royal Society, not to publish the research. Horton said a member of the Royal Society had called him and threatened him with the loss of his job if he did publish it.
Prof Howard reminded the audience of the pressure on the scientific establishment to suppress inconvenient findings. In 1999 Richard Horton, editor of The Lancet, told the Guardian that he had been pressured from "all quarters", notably from the UK Royal Society, not to publish the research. Horton said a member of the Royal Society had called him and threatened him with the loss of his job if he did publish it.
The Guardian identified the man as Peter Lachmann, former vice-president and biological secretary of the Royal Society and president of the Academy of Medical Sciences. The newspaper also drew attention to Lachmann's links to biotech companies.
In an editorial in The Lancet, Horton warned policymakers about the manipulation of scientific data by corporations and "of scientific colleagues being seduced by the material charms of industry. Trust is no defence against an aggressively deceptive corporate sector."
In conclusion, Prof Howard suggested a rigorous format for risk assessments, including identifying and modelling all hazards and being honest about areas of uncertainty. Assessors must state the levels of confidence in the results and the time scale over which the risk assessment can be considered valid. Hazard assessments should be fully transparent. Strict liability should be established and only temporary licences granted to a product or technology.
Prof Howard said society as a whole should decide whether certain existing products or technologies should be developed or continued. The onus should be on the developers to prove they are safe and risk assessment should be used only when appropriate. That's because, in Prof Howard's words, "No risk is acceptable if it is avoidable".
* GMWatch cautions that possible exceptions include:
– potential allergic reactions to Starlink GM maize, which was only approved for animal feed but got into the human food supply. More than 40 people reported allergic reactions and three people died, though Starlink was not proven to be the cause.
– the 37 deaths and 1500 cases of illness attributed to a food supplement, l-tryptophan, that was manufactured from GM bacteria engineered to ramp up production.










