EFSA ignored evidence of harm in GM-fed rats in spite of warnings from EU member state experts
A Monsanto genetically modified (GMO) insecticidal maize that has been approved for food and feed use in Europe for seven years was found to cause kidney disease and bladder stones in rats in industry’s own tests. Before the maize was approved, several EU member states raised concerns about these results and the implications for food safety. But the European Food Safety Authority (EFSA) dismissed the concerns and gave a favourable opinion on the GMO maize.[1] With the usual lack of agreement from EU member states on whether to authorize the maize, the Commission approved[2] it in 2011.
Since then, the GM trait associated with these adverse effects, known as MON89034, has been crossed with other GM maize varieties to form “stacked trait” GM crops containing multiple GM traits. As each new stacked trait GMO has come up for approval, member states have continued to draw attention to the adverse health impacts in the rats fed on MON89034 in the original feeding studies. France in particular has repeatedly raised concerns about the kidney and bladder effects in rats fed MON89034.
But EFSA has continued to downplay these experimental findings. It has issued favourable opinions that have led to the Commission taking the decision to approve nine stacked trait maize varieties containing MON89034.[3]
What’s in MON89034 maize?
MON89034, marketed as YieldGard™ VT Pro™, is a Monsanto GM maize that expresses its own Bt toxin insecticides. MON89034 maize contains a unique mix of insecticidal proteins called Bt toxins. The plants produce a synthetic Bt toxin, Cry1A.105 – a combination of Bt toxins called Cry1Ac/Cry1Ab and Cry1F. There is no natural form of this combined protein, so safety cannot be concluded by comparison with natural Bt toxins used previously. This synthetic toxin is combined with another Bt toxin, Cry2Ab2.[4]
Kidney and bladder adverse effects
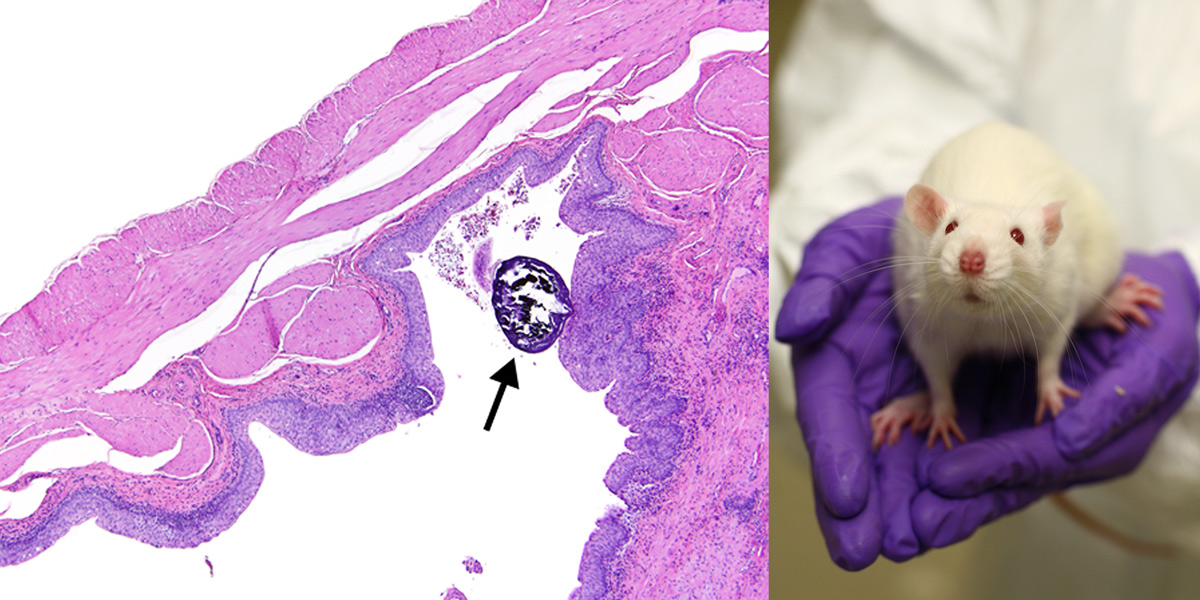
EFSA asserted that this GM maize was as safe as non-GMO maize in spite of findings in Monsanto’s own studies that two out of 20 female rats that ate the high dose of GM maize (33% of the total diet) developed kidney damage and bladder stones. One of the two rats died after just two weeks on the GM diet.
EFSA reported that the changes in the two rats included “minimal” chronic progressive kidney disease or damage, minimal to moderate transitional cell hyperplasia (cell proliferation that can be a precursor to cancer of the urinary system), inflammation, and hydronephrosis (presence of water in the kidneys due to obstruction). EFSA added that both rats had “urinary bladder calculi” – bladder stones. The study pathologist concluded that the tissue abnormalities observed were probably linked to the stones.
EFSA’s response
Astonishingly, EFSA convinced itself that the kidney damage and bladder stones in the GM maize-fed rats were a “spontaneous” or chance finding that had nothing to do with the GM diet. This was because, according to EFSA, “a low incidence of urinary bladder calculi [stones] is known to occur in this rat strain”.
EFSA added, “According to historical control data supplied in the application, the incidence of urinary bladder calculi in high dose females in this study was also found in female control rats in previous studies conducted with CD rats in the same testing laboratory. The [GMO] Panel therefore considers the urinary bladder calculi as well as the associated kidney alterations as incidental findings which were not related to administration of maize MON89034” (our emphasis).
Member states concerned
EFSA issued its favourable opinion on MON89034 in 2008, concluding that it was as safe as non-GMO maize.[1] But some EU member states strongly disagreed, as is clear from their comments on Monsanto’s application dossier in the three-month consultation period in 2007, leading up to the publication of EFSA’s opinion.[5]
Germany commented on the kidney and bladder results: “While the applicant suggests that these findings are unrelated to consumption of the test diet, possible adverse effects of MON89034 maize on the health of the test animals cannot be excluded from this result and from other significantly different nephrological [kidney] and haematological [blood] findings in MON89034 maize fed female rats.”[5]
The German experts, from the German Federal Agency for Nature Conservation (BfN), recommended that Monsanto be required to carry out an additional two-generation study with the maize to test for long-term health effects. But EFSA blandly replied that it saw “no reason to request additional feeding studies”.[5]
Austria also voiced concerns about the kidney and bladder findings, as did Belgium. The experts from Belgium’s Biosafety Advisory Council focused on an additional worrying finding: a low thyroid/parathyroid weight relative to final body weight was found in this same group of female rats fed a 33% GM maize diet. Referring to the whole roster of effects in the GM-fed rats, Belgium recommended having “a closer look” to see “whether these findings are solely due to chance. In case any doubt remains, further testing is recommended.”[5]
Historical control data
As we have seen above, EFSA repeatedly dismissed member states’ concerns about the bladder stones in the GM maize-fed female rats by referring to historical data that Monsanto had submitted from control group rats in other experiments. These data are unpublished and so cannot be checked by independent scientists. EFSA claimed that these data show that in this strain of rat, bladder stones or urinary calculi “sometimes appear”.[5] The implication was that MON89034 did not cause the problems.
However, the French experts pointed out a problem with this argument. The percentage of high-dose GM maize-fed rats that developed bladder stones was 10% of the group, compared with a relatively low background rate of 0.49% in the historical control data.[5] This is a huge discrepancy, which EFSA fails to mention or address in its opinion.
The French experts commented that “additional explanations” were required to explain the discrepancy. In the absence of this information, they reported, the French food safety agency ANSES “cannot conclude on the safety of products derived from maize varieties carrying the MON89034 transformation event”.[5]
The French were not reassured by Monsanto’s historical control data, as shown by a later opinion[6] issued in 2016 by ANSES on Monsanto’s application for the approval of a multi-trait (stacked trait) GM maize containing the MON89034 trait, called MIR162 x MON87427 x MON89034 x NK603.
ANSES stated: “Although historical data from 70 studies conducted between 1999 and 2006 with rats of the same strain were provided by the petitioner, they did not appear sufficient to conclude that there was no link between the oral administration of MON89034 maize and the occurrence of bladder stones observed in female animals fed the high dose of MON89034.”[6]
ANSES repeated these concerns in a 2017 opinion on another stacked trait maize, MON89034 x 1507 x NK603 x DAS-40278-9.[7]
Risks stack up
As is clear from ANSES’s recent opinions cited above, EU member states’ concerns over MON89034 have not gone away. Rather they have intensified as this GM Bt maize variety has been crossed with other GM Bt maize varieties to produce “stacked” multi-GM-trait crops. The database run by the industry body ISAAA lists a total of 10 GMO maize varieties containing the MON89034 trait that have been approved in Europe for food and feed use.[8]
With stacked trait GM crops, there is a risk that the multiple GMO components of the crop will interact with each other, with other elements in the plant, or with the agrochemicals applied during cultivation, and create new or enhanced toxins or allergens.
In spite of these heightened risks, EFSA does not assess the combined effects of the different GM traits in a stacked variety, nor does it require a 90-day animal toxicity feeding study with the stacked GM crop, such as is required for single-trait GM varieties. Instead EFSA bases its safety assessment on the previous animal feeding studies with the single-trait GM varieties that were crossed to make the stacked-trait variety.[9,10]
Shaky foundation
In 2007 several member states expressed concerns about the safety of a stacked trait GM maize variety containing MON89034, known as MON89034 x MON88017.[11] But EFSA failed to respond to any of these concerns with hard evidence or substantive scientific argument and in 2011 MON89034 x MON88017 was approved for food and feed use.[12]
In a comment that questions whether a solid structure can be built on a shaky foundation, Austria stated that EFSA’s risk assessments of the single traits that went into MON89034 x MON88017 were “insufficient”, so considering an approval for the stacked crop before questions about the single traits had been clarified was not “appropriate”.[11]
Austria also challenged the need for this GM crop by stating that the rootworm pest targeted by the Bt toxins in the GM maize can be controlled by simple crop rotations. It also pointed out that many components of the GM maize were statistically significantly different from the controls and recommended molecular profiling “omics” analysis to detect unintended effects.[11]
France, Austria, and Belgium worried that no animal feeding toxicology studies with the complete stacked trait GM crop had been done, so the safety of the combined Bt toxins had not been tested.[11]
Austria stated, “Despite the new risk of interactions based on gene-stacking no toxicity feeding studies were conducted. The studies carried out on the single events are not regarded as sufficient for the risk assessment of novel constituents and proteins by stacking.”[11]
Belgium stated that a 13-week feeding study in rats should be performed with the stacked trait crop since “synergistic effects” of the combined proteins in the maize “cannot be excluded”.[11]
Allergen worries
Belgium worried about the allergenicity of the synthetic Cry1A.105 protein since it shares a 24% similarity with the major allergen in kiwi fruit. The Belgian delegation recommended performing skin tests with the Cry1A.105 protein on kiwi-sensitized patients, as well as post-market surveillance for adverse reactions. EFSA dismissed this idea on the grounds that a threshold of 35% similarity with a known allergen is not reached. But Belgium had already rejected this argument, giving detailed technical reasons why the 35% threshold may not be adequate to spot allergens in this GMO.[11]
Austria complained about the design of a feeding test on 600 broiler chickens, in which only 100 birds were fed the GM maize.[11]
This faulty design, common in industry animal feeding studies on GMOs, leads to a situation in which any changes seen in the GM-fed birds would disappear among the wide range of values obtained from the 500 non-GM-fed birds, making the experiment so insensitive as to be worthless as a test of feed safety. Indeed, EFSA defended the test by saying that it is not meant to address toxicity but only the nutritional equivalence of the GM and non-GM maize varieties. That’s all very well, but EFSA’s response did not address the fundamental issue that a proper toxicological feeding study with the stacked trait crop had not been done.
EFSA misses the point
France reminded EFSA about the bladder stones and kidney effects in rats that ate MON89034 in Monsanto’s original feeding study. The French experts commented that it was not possible to conclude from these data that products derived from MON89034 x MON88017 maize are not toxic. They added that a 90-day feeding study should be carried out with the stacked trait crop to ensure that these changes are not due to “genetic modification introduced into MON89034 maize”.[11]
EFSA replied that it had previously found the single events to be safe to eat and added that “neither the molecular structures of MON 89034 x MON88017 nor the protein expression levels have been found to be changed in comparison to the single events in the analysis of molecular characteristics”. The agency added that it does not consider it “necessary” to request an animal feeding study with the stacked trait plant.[11]
However, for Dr Michael Antoniou, a London-based molecular geneticist, EFSA is missing (or avoiding) the point made by the French experts.
Dr Antoniou said: “The issue is not whether the molecular structures or the protein expression levels have changed, but whether Bt toxins combined with one another or with natural compounds in the plant have biological effects. EFSA seems to be saying that the applicant has measured the levels of individual Bt toxins in the stacked trait plant and found them to be no different from the levels in the single-trait plants. However, that is not what the French are questioning. They want to know if the combinations of Bt toxins and/or mixtures of Bt toxins with natural plant compounds might produce an additive or synergistic effect that cannot be predicted from looking at the Bt toxins individually.
“The stacked trait crop needs to be tested in its own right. It is not scientifically valid to extrapolate data from single GM traits to a stack. To do so leads to unsubstantiated conclusions. The only way to answer questions about the safety of the stacked trait crop is to do a long-term animal feeding experiment. In asserting that an animal feeding study with MON89034 x MON88017 is not needed, EFSA is expressing an opinion rather than making an evidence-based statement.
“In EFSA’s view, if it concludes that there is no harm from single traits, there is also no harm when they are combined. But we know from studies on chemical pollutants that substances tested individually at certain doses may show no ill effects, but when combined at the same concentrations, suddenly they show severe adverse effects.[13] The same could happen with Bt toxins. But until the experiments are done, no one knows.”
Dr Antoniou’s concerns are primarily with mammalian and human health effects, where there is a dearth of studies on the combined effects of Bt toxins. But a recently published scientific review found a number of studies on insects – both those targeted by the Bt toxins and “non-target” and beneficial insects – confirming that interactions and combined effects do occur between Bt toxins or Bt toxins and other proteins, chemicals, soil organisms, or plant components, in some cases resulting in unexpected increases in toxicity. The mechanisms of these effects are not fully understood.[14,15]
Crucially, in a finding that directly challenges EFSA’s reductionist “one-at-a-time” approach to stacked-trait crops, the authors of the review discovered that many synergistic interactions in target organisms were not predictable from single trait effects. They occurred when the individual components tested in isolation did not elicit a response at all or elicited only a non-lethal response.
“No scientific knowledge” on combination effects
In the case of the stacked GM Bt and herbicide-tolerant maize known as SmartStax,[16] which contains the MON89034 trait, produces six different Bt toxins, and is tolerant to two herbicides, EFSA stated, without referencing any experimental evidence, that there were no safety concerns about possible combined effects.[17]
Experts from EU member states such as Austria, Belgium and Germany disagreed. They emphasized the need to carry out feeding studies to test for combined and synergistic effects. The Austrian experts stated: “The safety of all newly expressed proteins in animal models applied simultaneously and combined was not assessed in the dossier. Insecticidal Cry proteins produced by GM plants as well as transproteins conferring tolerance to herbicides constitute a sum of new plant constituents possibly interacting within the organism. So far, there is absolutely no scientific knowledge about such those in the respective new combinations and possibly resulting additive and/or synergistic effects.”[18]
Nonetheless, the EU Commission approved SmartStax for food and feed use in 2013.[19]
Mistrust
The story of MON89034 maize shows why some EU member states and the public do not trust EFSA. The agency has repeatedly failed to address scientific concerns about this GM trait with hard evidence. Notably, it has been unable to convince skeptical member states of its view that an increased rate of bladder stones and kidney damage in female rats fed with MON89034 were due to chance and nothing to do with the GM maize diet. EFSA cited unpublished historical control data submitted by Monsanto to support its claim but failed to address the obvious problem of the massive increase in the rate of bladder stones in MON89034-fed female rats (20%) over the historical control data rate (0.49%).
MON89034 has now been incorporated into stacked trait GM crops. Thus the risk associated with this GM trait has been augmented by the risks from combined GM traits, along with the risks posed by interactions of these traits with natural plant compounds or soil organisms. Unsurprisingly, member states that were not convinced of the safety of MON89034 are doubly skeptical of the safety of stacked trait crops – which they believe are already inadequately regulated – containing the MON89034 trait.
As GM crops become more complex, skeptical EU member states and the public are likely to grow more mistrustful of EFSA’s failures to address risk and of the Commission’s actions in approving these crops without a democratic mandate.
Report: Claire Robinson
References
1. EFSA (2008). Application (Reference EFSA-GMO-NL-2007-37) for the placing on the market of the insect-resistant genetically modified maize MON89034, for food and feed uses, import and processing under Regulation (EC) No 1829/2003 from Monsanto[1]. EFSA Journal 909:1-30. https://www.efsa.europa.eu/en/efsajournal/pub/909
2. EU Commission (2011). COMMISSION DECISION of 17 June 2011 authorising the placing on the market of products containing, consisting of, or produced from genetically modified maize MON 89034 × MON 88017 (MON-89Ø34-3xMON-88Ø1 7-3) pursuant to Regulation (EC) No 1829/2003 of the European Parliament and of the Council. http://bit.ly/2suATwS
3. ISAAA (2018). GM crop events approved in European Union. http://www.isaaa.org/gmapprovaldatabase/approvedeventsin/default.asp?CountryID=EU
4. Testbiotech and Genewatch UK (undated). Request for internal review of the market authorisation for genetically engineered maize MON80934 x MON88017, Monsanto, Genuity VT Triple PRO Corn, under Article 10 of Regulation (EC) No. 1367/2006. http://bit.ly/2Eqe9zv
5. EFSA (undated). Application EFSA-GMO-NL-2007-37 (MON89034 Maize). Annex G. Comments and opinions submitted by Member States during the three-month consultation period. http://bit.ly/2BWhybG
6. ANSES (2016). Saisine no. 2016-SA-0143: Avis de l'Agence nationale de sécurité sanitaire de l'alimentation, de l'environnement et du travail relatif à une demande d'autorisation de mise sur le marché, au titre du Règlement (CE) no. 1829/2003 relatif aux denrées et aux aliments pour animaux génétiquement modifiés, du maïs génétiquement modifié MIR162 x MON87427 x MON89034 x NK603, développé pour être résistant à certains insectes et tolérant au glyphosate, pour l'importation, la transformation ainsi que l'utilisation en alimentation humaine et animale de cet OGM (dossier no. EFSA-GMO-NL-2016-131). 8 August. https://www.anses.fr/fr/system/files/BIOT2016SA0143.pdf
7. ANSES (2017). Saisine no. 2016-SA-0244: Avis de l'Agence nationale de sécurité sanitaire de l'alimentation, de l'environnement et du travail relatif à une demande d'autorisation de mise sur le marché, au titre du Règlement (CE) no. 1829/2003, du maïs génétiquement modifié MON89034 x 1507 x NK603 x DAS-40278-9, développé pour être résistant à certains insectes et tolérant à plusieurs herbicides (glyphosate, glufosinate-ammonium, 2,4-D et herbicides de la famille des aryloxyphénoxypropionates (AOPP)), pour l'importation, la transformation ainsi que l'utilisation en alimentation humaine et animale de cet OGM (dossier no. EFSA-GMO-NL- 2013-112). 10 February. https://www.anses.fr/fr/system/files/BIOT2016SA0244.pdf
8. ISAAA (2018). GM crop events approved in European Union. http://www.isaaa.org/gmapprovaldatabase/approvedeventsin/default.asp?CountryID=EU
9. Then C and Bauer-Panskus (2011). “...ensured that the data were consistent with expectations...”: How industry and EFSA have been systematically undermining the risk assessment of “SmartStax”. Testbiotech. https://www.testbiotech.org/sites/default/files/Testbiotech_SmartStax_en.pdf
10. GMWatch (2014). Stacking traits in a GMO is found to cause unexpected synergistic effects. 17 Dec. http://www.gmwatch.org/en/news/archive/2014/15820
11. EFSA (undated). Application EFSA-GMO-NL-2007-39 (Maize MON 89034 x MON 88017): Comments and opinions submitted by Member States during the three-month consultation period. http://registerofquestions.efsa.europa.eu/roqFrontend/wicket/page?1-2.ILinkListener-buttonform-questionDetailsTabs-panel-docItemForm-pageable-10-fileNameLnk
12. ISAAA (2016). Event Name: MON89034 x MON88017. http://www.isaaa.org/gmapprovaldatabase/event/default.asp?EventID=96
13. Rajapakse N et al (2002). Combining xenoestrogens at levels below individual no-observed-effect concentrations dramatically enhances steroid hormone action. Environ Health Perspect 110(9):917–921. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1240992/
14. Hilbeck A and Otto M (2015). Review article: Specificity and combinatorial Effects of Bacillus thuringiensis Cry toxins in the Context of GMO environmental risk assessment. Front. Environ. Sci. 9 November. http://dx.doi.org/10.3389/fenvs.2015.00071
15. Robinson C (2015). Scientists warn of dangers of multi-toxin Bt crops. GMWatch, 7 Dec. http://www.gmwatch.org/en/news/latest-news/16583
16. MON89034 x 1507 x MON88017 x 59122.
17. EFSA (2017). Scientific Opinion on application EFSA-GMO-BE-2013-118 for authorisation of genetically modified maize MON 87427 x MON 89034 x 1507 x MON 88017 x 59122 and subcombinations independently of their origin, for food and feed uses, import and processing submitted under Regulation (EC) No 1829/2003 by Monsanto Company. EFSA Journal 15(8):4921. http://onlinelibrary.wiley.com/doi/10.2903/j.efsa.2017.4921/epdf
18. “ensured that the data were consistent with expectations...”: How industry and EFSA have been systematically undermining the risk assessment of “SmartStax”. Testbiotech. https://www.testbiotech.org/sites/default/files/Testbiotech_SmartStax_en.pdf
19. ISAAA (2017). Event Name: MON89034 x TC1507 x MON88017 x 59122. http://www.isaaa.org/gmapprovaldatabase/event/default.asp?EventID=121
The photo of bladder stone in a rat on the left is from the US National Toxicology Program; it does not relate to the rat feeding study with MON89034 featured in this article. The photo of an experimental rat on the right is from Understanding Animal Research.










